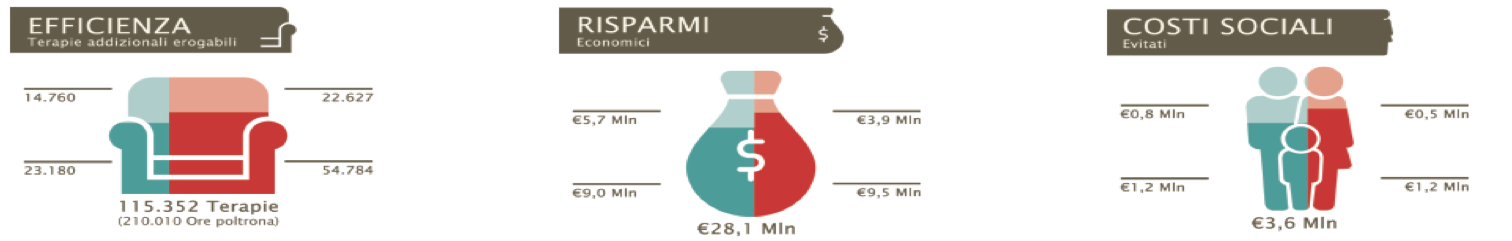
Objective
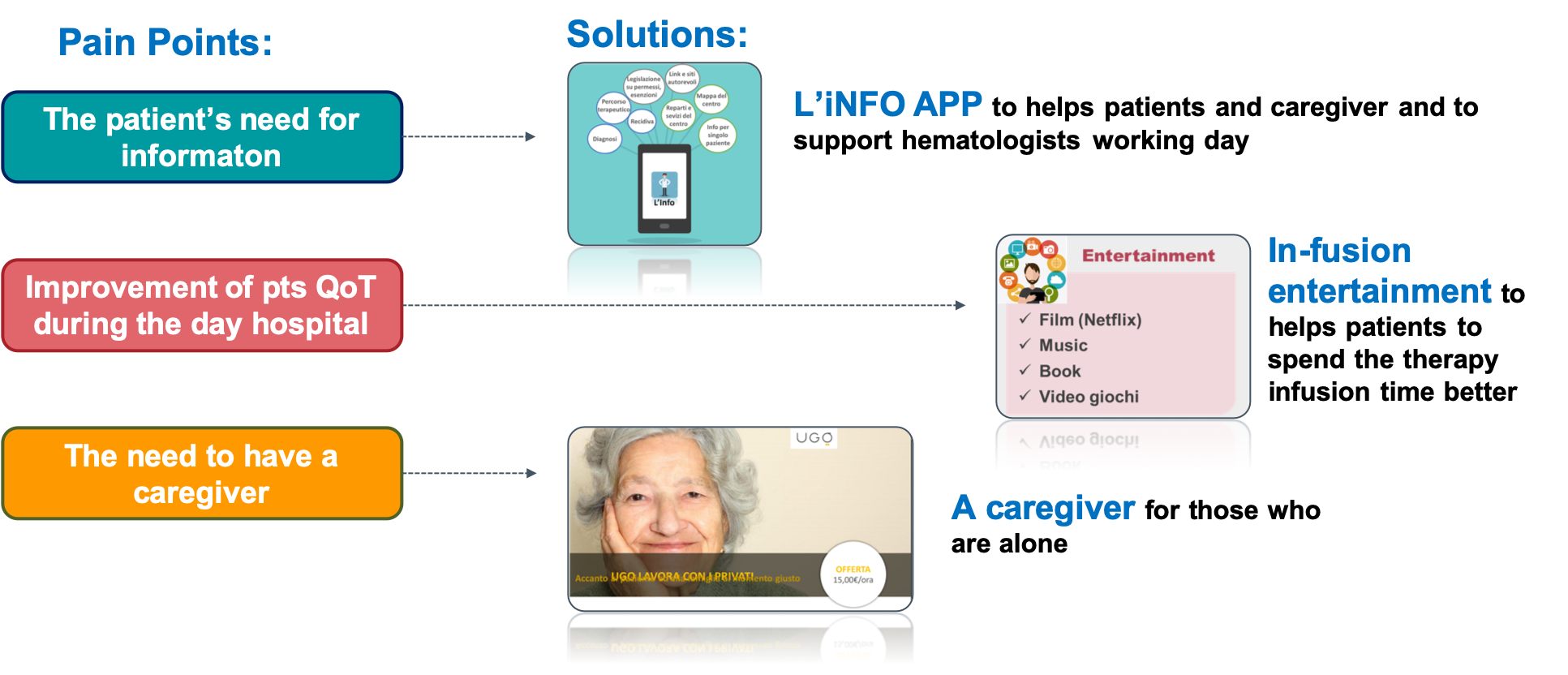
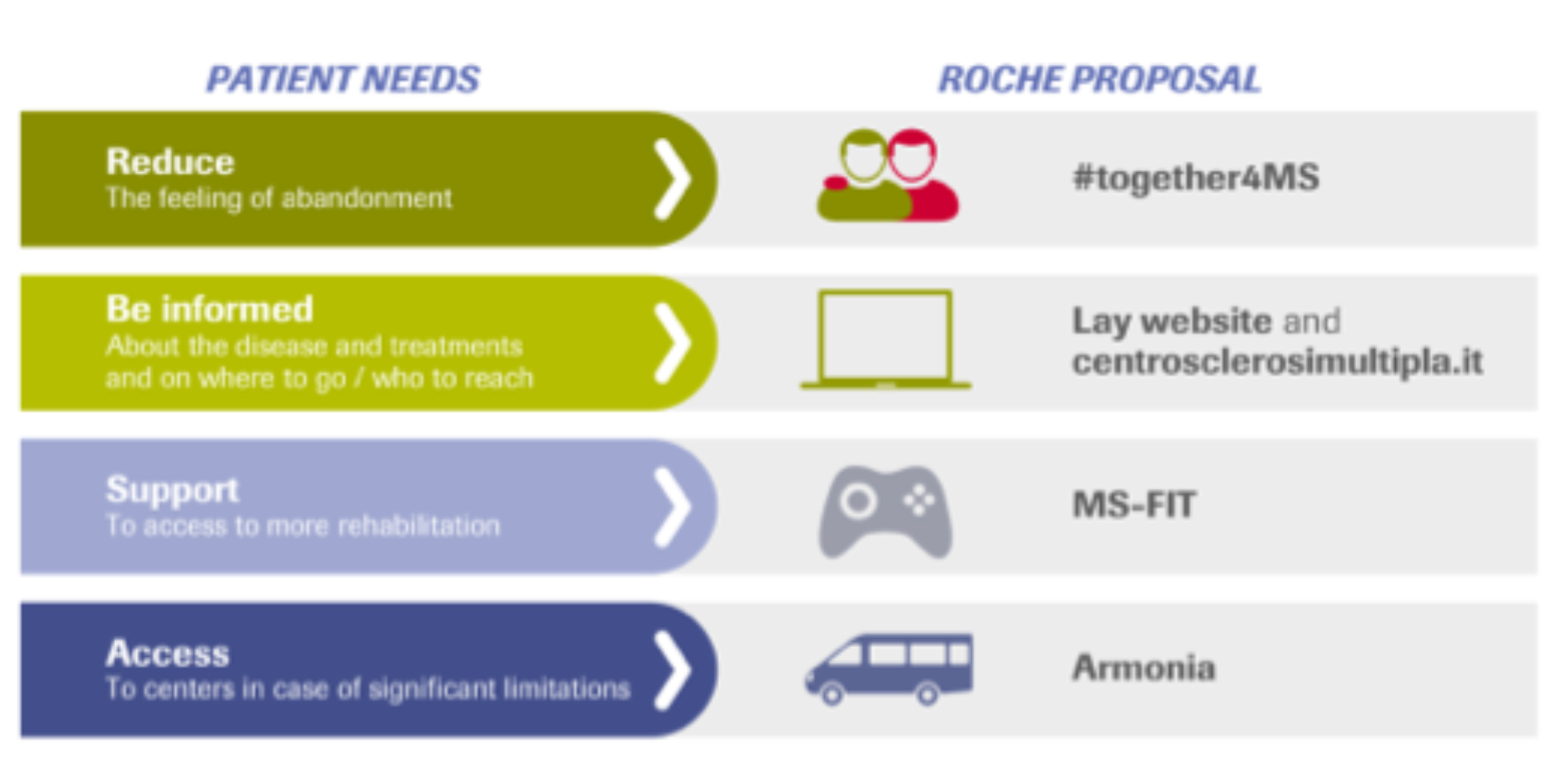
Armonia Project, which prepared and supported the Ocrevus launch, aimed at satisfy unmet needs of the MS community offering our customers target services in addition to Ocrevus, implementing a strategy beyond the product.
The main objective I pursued within the project was the identification of the best legal solutions to make the additional services available to our customers in conjunction with the product, exploring innovative and more collaborative approaches different than traditional ones, like donations, and shaping the legal environment in this sense.
The Armonia experience guided us to a new market access and commercial mindset, now in course of definition with sprint sessions, in order to define a business strategy based on the systematic integration between products and services as a preliminary basis of our market access and commercial offers.
Export/Import
The Italian experience has been presented within the global network (with yearly follow up), sharing our practice, our commercial focus and our deep connection with the legislative framework.
We annually report the most relevant learnings, based on solutions that were successfully implemented, feasibility barriers and obstacles from the customers’ perspective.
Outcome
Despite some initial reluctances, we succeeded in shaping the pharma environment (also considering new draft of law under discussion on these topics), approaching customers with flexible implementation proposals, including both:
-
joint offer of products and services into commercial offers (according to regional tender notices admitting services in addition to drugs) and
-
targeted negotiations of public sponsorship agreements - instead of donations – with accounts and university, positioning Roche as a real partner instead of a pure payer.
Learning
Challenging a system traditionally based on a systematic routine of donations and not admitting alternatives is not easy.
Working in team, involving colleagues interacting with different customers, knowing deeply our customers’ governance and processes and flexible – targeted approaches are essential to accompany the customers into a new mindset and exploration of legislative opportunities not yet in use in the pharmaceutical sector.
Our north star must guide us in defining integrated products and services solutions, which are the best answers to our customers’ needs, not only in terms of outcome but also in terms of fast implementation and high level of transparency and legal compliance.






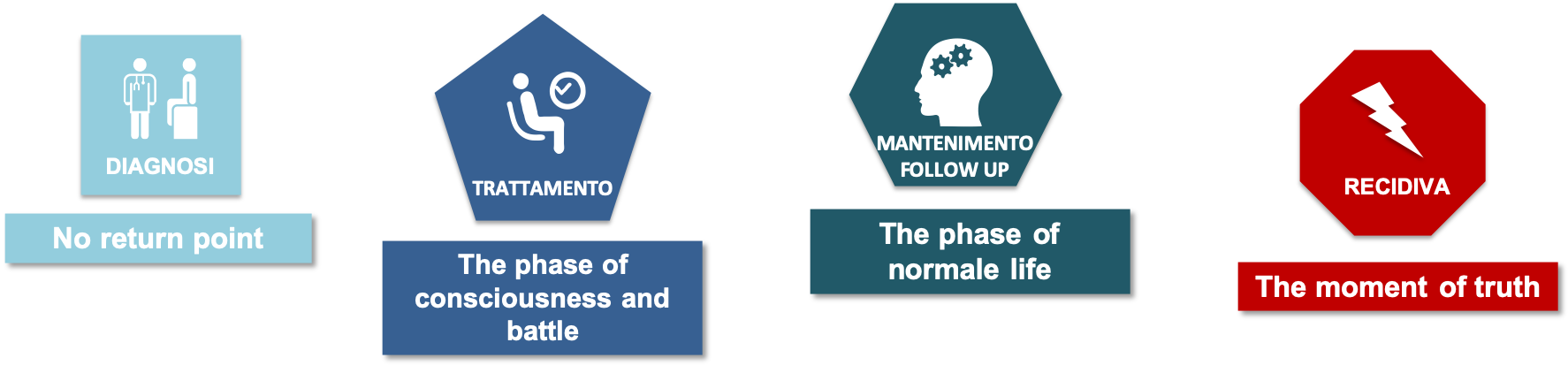
 The value for clinicians of projecting FL patients towards a similar or identical OS, to that of a control population, not affected by this disease.
The value for clinicians of projecting FL patients towards a similar or identical OS, to that of a control population, not affected by this disease.